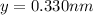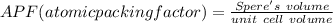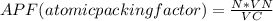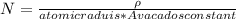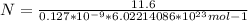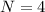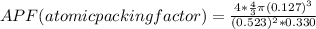Chemistry, 06.02.2021 05:40 91miketaylor
Consider a hypothetical metal that has a density of 11.6 g/cm3, an atomic weight of 157.6 g/mol, and an atomic radius of 0.127 nm. Compute the atomic packing factor if the unit cell has tetragonal symmetry; values for the and lattice parameters are 0.523 nm and 0.330 nm, respectively.

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 07:40, caleb19moody
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2
You know the right answer?
Consider a hypothetical metal that has a density of 11.6 g/cm3, an atomic weight of 157.6 g/mol, and...
Questions in other subjects:


Chemistry, 30.01.2021 14:00

Mathematics, 30.01.2021 14:00


Mathematics, 30.01.2021 14:00

English, 30.01.2021 14:00

Mathematics, 30.01.2021 14:00


Mathematics, 30.01.2021 14:00

Mathematics, 30.01.2021 14:00

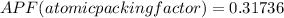
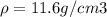
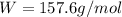
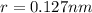
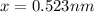 and
and