
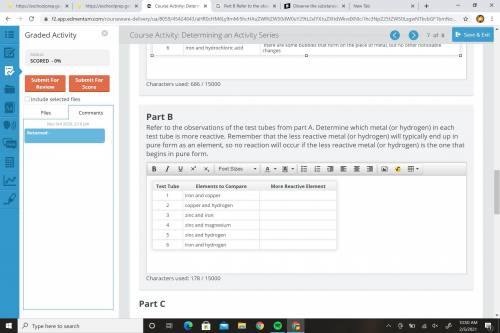
Please help me... Refer to the observations of the test tubes from part A. Determine which metal (or hydrogen) in each test tube is more reactive. Remember that the less reactive metal (or hydrogen) will typically end up in pure form as an element, so no reaction will occur if the less reactive metal (or hydrogen) is the one that begins in pure form.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, britotellerialuis
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1


You know the right answer?
Please help me...
Refer to the observations of the test tubes from part A. Determine which metal (...
Questions in other subjects:

Mathematics, 25.08.2019 18:30

Chemistry, 25.08.2019 18:30





Mathematics, 25.08.2019 18:30

Mathematics, 25.08.2019 18:30

English, 25.08.2019 18:30



