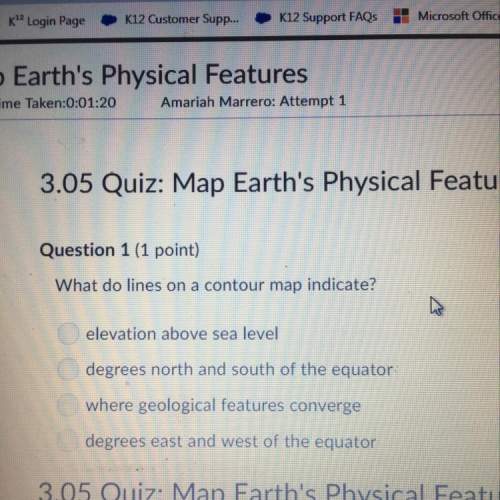
Chemistry, 05.02.2021 05:30 someone2301
Plot a graph of volume vs. temperature (in kelvins) with the two data points that resulted from your experiment. Draw a straight line connecting the two points, thus assuming a linear relationship between volume and temperature. The x-intercept corresponds to where the volume would be zero and the temperature can be no lower, defined as absolute zero. Absolute zero is 0 K. Compare your results to those expected. How close to absolute zero was your intercept? Why might your value be different from absolute zero?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, krharris
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 10:00, JOEFRESH10
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 22.06.2019 14:00, njones58emailtjcedu
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1
You know the right answer?
Plot a graph of volume vs. temperature (in kelvins) with the two data points that resulted from your...
Questions in other subjects:


English, 05.11.2020 14:00


History, 05.11.2020 14:00

Mathematics, 05.11.2020 14:00

History, 05.11.2020 14:00


Mathematics, 05.11.2020 14:00