Chemistry, 02.02.2021 20:30 sunshine52577oyeor9
3. A sample of neon gas has a volume of 1.2 L at 28.5°C. What will the volume be at 125°C it the pressure remains constant?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, melissa9882
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 21:00, Janznznz4012
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3

Chemistry, 23.06.2019 00:00, glocurlsprinces
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3

Chemistry, 23.06.2019 03:00, andrejr0330jr
Which of the following is a chemical property of water at 4 c
Answers: 2
You know the right answer?
3. A sample of neon gas has a volume of 1.2 L at 28.5°C. What will the volume be at 125°C it the
pr...
Questions in other subjects:

Mathematics, 20.07.2019 04:10

Mathematics, 20.07.2019 04:10

Mathematics, 20.07.2019 04:10







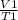 =
= 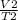
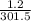 =
=