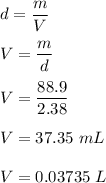Chemistry, 02.02.2021 09:50 janeekajones08
What volume, in liters, would 88.9 grams of a substance occupy if it’s density is 2.38 g/mL

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 11:30, ashleybarrera2000
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Chemistry, 22.06.2019 16:30, joshua1255
Find the number of moles of argon in 364g of argon.
Answers: 2

Chemistry, 23.06.2019 05:00, MoneyMike42
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3
You know the right answer?
What volume, in liters, would 88.9 grams of a substance occupy if it’s density is 2.38 g/mL...
Questions in other subjects:


Mathematics, 16.12.2020 21:00


English, 16.12.2020 21:00

Mathematics, 16.12.2020 21:00



Health, 16.12.2020 21:00