
Chemistry, 01.02.2021 18:20 allycoops666666
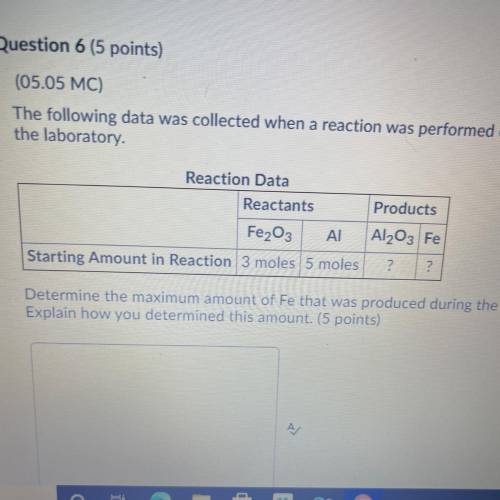
The following data was collected when a reaction was performed experimentally in the laboratory. Determine the maximum amount of Fe that was produced during the experiment. Explain how you determined this amount.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 13:00, carlinryan
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2
You know the right answer?
The following data was collected when a reaction was performed experimentally in the laboratory. De...
Questions in other subjects:

English, 24.02.2020 09:43


English, 24.02.2020 09:43

Health, 24.02.2020 09:43

Biology, 24.02.2020 09:44

Chemistry, 24.02.2020 09:44



Biology, 24.02.2020 09:46

Business, 24.02.2020 09:46



