FILL OUT THE GRAPH AND ANSWER THE QUESTIONS WILL MARK BRAINLIEST
PIC OF GRAPH ATTACHED
...

FILL OUT THE GRAPH AND ANSWER THE QUESTIONS WILL MARK BRAINLIEST
PIC OF GRAPH ATTACHED
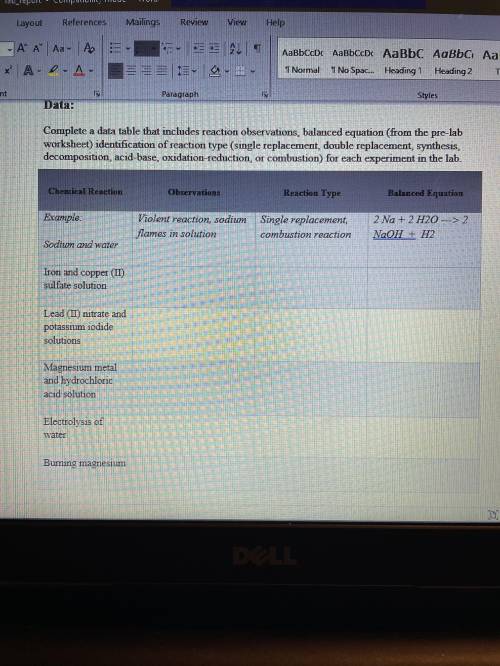
Conclusion:
Write a conclusion statement that addresses the following questions:
• Summarize your observations of each reaction. Based on the products made, were your predictions of chemical reaction type correct?
• Some of the reactions you conducted can be categorized as more than one type of reaction. Which reactions are these, and what are the types of reactions?
• How do you think the investigation can be explored further?
Post-Lab Reflection Questions
Answer the reflection questions using what you have learned from the lesson and your experimental data. It will be helpful to refer to your chemistry journal notes. Answer questions in complete sentences.
1. If you were to measure the mass of magnesium and hydrochloric acid before combining them in the test tube, how would that mass compare to the mass of reactants left in the test tube after the reaction? Explain your answer and how it corresponds to the law of conservation of mass.
2. In what other ways could you test your predictions to confirm their accuracy?
3. How do the chemical reactions in this lab activity compare to nuclear reactions, such as fission and fusion?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, isaiahrodriguezsm17
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1

Chemistry, 22.06.2019 16:00, sassy11111515
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1

Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
Questions in other subjects:












