
Chemistry, 27.01.2021 01:00 kelseybieberrr19
It would likely be most difficult to collect a sample of which isotope?
Pb-206
Po-214
U-234
Ra-226
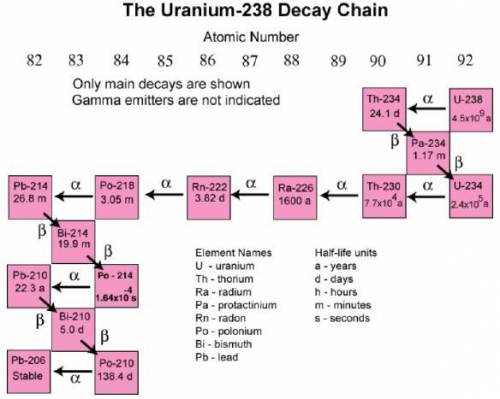

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 16:00, winnie45
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 22:30, brianna5626
How do limiting factors most affect population size? ostop population growthrestrict population growthincrease population sizeresult in positive impactso
Answers: 1
You know the right answer?
It would likely be most difficult to collect a sample of which isotope?
Pb-206
Po-214
U...
Po-214
U...
Questions in other subjects:


Health, 26.05.2020 03:01

History, 26.05.2020 03:01

Chemistry, 26.05.2020 03:01


History, 26.05.2020 03:01




English, 26.05.2020 03:01



