The kp for the reaction below is 1.49 × 108 at 100.0°c:
co(g) + cl2(g) → cocl2(g)
...

The kp for the reaction below is 1.49 × 108 at 100.0°c:
co(g) + cl2(g) → cocl2(g)
in an equilibrium mixture of the three gases, pco = pcl2 = 2.22 × 10-4 atm. the partial pressure of the product, phosgene (cocl2), is atm.
a) 7.34
b) 3.02 × 10^15
c) 3.31 × 10^-16
d) 3.31 × 10^4
e) 6.67 × 10^11

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, shadekashakay
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 10:00, paynedeforest2596
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 13:50, hannahmyung1113
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3

Chemistry, 22.06.2019 14:30, Tooey2331
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3
You know the right answer?
Questions in other subjects:





Computers and Technology, 09.12.2019 20:31

English, 09.12.2019 20:31




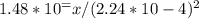 . So, the correct answer is A) 7.34.
. So, the correct answer is A) 7.34.

