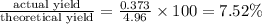Chemistry, 25.01.2021 18:00 MikeCrotch19251
2H2 (1) + O2(g) → 2H20 (g)
1. Find the limiting reactant if you start with 30.0 grams of hydrogen and 5.29 grams of oxygen.
2. The actual yield for H2O in the above reaction is 6.72 g, Determine the percent yield for the reaction
when 9.93 grams of hydrogen and excess oxygen react?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:40, hardwick744
Achange in the number of neutrons in an atom will change an blank . when the number of protons changes in an atom, a new element will form.
Answers: 2



Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
2H2 (1) + O2(g) → 2H20 (g)
1. Find the limiting reactant if you start with 30.0 grams of hydrogen a...
Questions in other subjects:

Mathematics, 07.01.2021 01:10

Mathematics, 07.01.2021 01:10

Mathematics, 07.01.2021 01:10



English, 07.01.2021 01:10


Mathematics, 07.01.2021 01:10


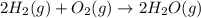
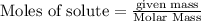
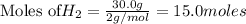
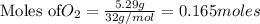
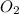 require = 2 moles of
require = 2 moles of 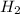
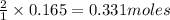 of
of 
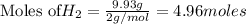
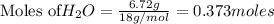

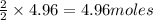 of
of