
Chemistry, 25.01.2021 05:40 gracerhodes5364
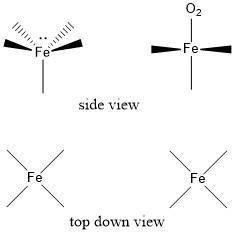
Critical thinking problem: The iron in hemoglobin in our red blood cells is used to bind and carry dioxygen throughout the body. Iron is one of the atoms that can form hybrid orbitals using its d-shell electrons, which allows for a greater number of sigma bonds than just four. When iron is only bound to protein, it forms five sigma bonds with one lone pair. The iron atom is pulled down below the central square plane due to one of the bonds with the hemoglobin protein. The thick lines indicate bonds pointing out of the screen, while dashed lines indicate bonds pointing into the screen, so as to suggest 3-D structure.
When iron binds to dioxygen, it now has six sigma bonds. The geometry changes, even though the hybridization orbitals do not change. The dashed lines are still present, as seen from the top-down view, but the thick lines hide them in the side-on view.
Why would the molecular geometry for iron change in this case? Would this have anything to do with the ability of hemoglobin to bind oxygen tightly?

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, cheesecake1919
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 11:50, trinityrae4657
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 23.06.2019 02:00, matthewsorrow02
What is the mass of 0.750 mole of aluminum oxide, al2o3?
Answers: 1
You know the right answer?
Critical thinking problem: The iron in hemoglobin in our red blood cells is used to bind and carry d...
Questions in other subjects:

Social Studies, 20.11.2019 09:31

Mathematics, 20.11.2019 09:31

History, 20.11.2019 09:31


Mathematics, 20.11.2019 09:31




Mathematics, 20.11.2019 09:31



