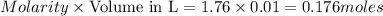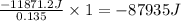Chemistry, 24.01.2021 14:00 shaheedbrown06
A reaction of 100mL of 1.35 M HCl and 100mL of 1.76 M NaOH is monitored and the following
temperatures were recorded: starting temperature = 24.6 C; and final temperature = 38.8 C.
Calculate the AH of this reaction.
A) -92142
J/Mole
B) -12439.2
J/Mole
C) 876
J/Mole

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 19:00, QuestionsAnsweredNow
Suppose that a certain fortunate person has a net worth of $71.0 billion ($7.10×1010). if her stock has a good year and gains $3.20 billion (3.20×109) in value, what is her new net worth?
Answers: 3
You know the right answer?
A reaction of 100mL of 1.35 M HCl and 100mL of 1.76 M NaOH is monitored and the following
temperatu...
Questions in other subjects:

Mathematics, 05.05.2021 23:40

Mathematics, 05.05.2021 23:40



Mathematics, 05.05.2021 23:40


Mathematics, 05.05.2021 23:40


Mathematics, 05.05.2021 23:40

Biology, 05.05.2021 23:40

 for the reaction is 87935 J
for the reaction is 87935 J
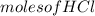 =
=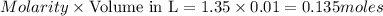
 =
=