
Chemistry, 21.01.2021 22:20 stephensandrew4293
When the concentration of A in the reaction A B was changed from 1.20 M to 0.60 M, the half-life increased from 2.0 min to 4.0 min at 25°C. Calculate the order of the reaction and the rate constant.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 23.06.2019 05:40, Queenquestion9130
Why is any chemical reaction always balanced? give reasons and explain the easiest way to solve the balancing problems in chemical equations with stoichiometric coefficients upto 20 as hit and trial doesn't always work. give full reasoning
Answers: 1

Chemistry, 23.06.2019 10:00, jdmXdude3140
How to draw a diagram to represent a calcium metal lattice?
Answers: 3

Chemistry, 23.06.2019 11:30, elizebeth4501
Which of these have the same number of particles as 1 mole of water h2o
Answers: 1

Chemistry, 23.06.2019 16:50, alexisbaronetp85kek
How can a scientist assess whether a pure niobium (nb) sample is responsible for contaminating the lab with radioactivity? test the niobium sample to see whether it now contains other elements. test the niobium sample for the presence of niobium oxide compounds. heat the niobium, and see if the level of radioactivity in the lab increases. place the niobium under pressure, and see if the level of radioactivity in the lab increases.
Answers: 3
You know the right answer?
When the concentration of A in the reaction A B was changed from 1.20 M to 0.60 M, the half-life in...
Questions in other subjects:

History, 05.11.2020 21:40

Arts, 05.11.2020 21:40

Mathematics, 05.11.2020 21:40

Health, 05.11.2020 21:40

Mathematics, 05.11.2020 21:40




Mathematics, 05.11.2020 21:40

Social Studies, 05.11.2020 21:40

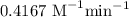
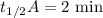
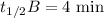
![{[A]_0}_A=1.2\ \text{M}](/tpl/images/1053/6371/ba19d.png)
![{[A]_0}_B=0.6\ \text{M}](/tpl/images/1053/6371/61cc0.png)
![t_{1/2}\propto \dfrac{1}{[A]_0^{n-1}}](/tpl/images/1053/6371/3ebb4.png)
![\dfrac{{t_{1/2}}_A}{{t_{1/2}}_B}=\left(\dfrac{{[A]_0}_B}{{[A]_0}_A}\right)^{n-1}\\\Rightarrow \dfrac{2}{4}=\left(\dfrac{0.6}{1.2}\right)^{n-1}\\\Rightarrow \dfrac{1}{2}=\left(\dfrac{1}{2}\right)^{n-1}](/tpl/images/1053/6371/86904.png)
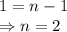
![t_{1/2}=\dfrac{1}{k[A]_0^{n-1}}\\\Rightarrow k=\dfrac{1}{t_{1/2}[A]_0^{n-1}}\\\Rightarrow k=\dfrac{1}{2\times 1.2^{2-1}}\\\Rightarrow k=0.4167\ \text{M}^{-1}\text{min}^{-1}](/tpl/images/1053/6371/f48ea.png)


