

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:10, hannacarroll2539
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1

Chemistry, 22.06.2019 21:30, Turtlelover05
How can the periodic table be used to predict the behavior of elements?
Answers: 1

Chemistry, 22.06.2019 22:30, medinajocelyn45
Which compound most likely has the greatest bond energy?
Answers: 2

Chemistry, 23.06.2019 09:40, ventalexander8406
Write balanced nuclear equations for the formation of five elements whose atomic number is between helium (2) and iron (26):
Answers: 1
You know the right answer?
Given 8.10g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesiz...
Questions in other subjects:



History, 17.12.2021 02:20







 butanoic acid = 0.09193 moles of butanoic acid
butanoic acid = 0.09193 moles of butanoic acid = 10.7 g
= 10.7 g of ethyl butyrate would be synthesized by 8.10 g of butanoic acid and excess ethanol.
of ethyl butyrate would be synthesized by 8.10 g of butanoic acid and excess ethanol.

 is as follows:
is as follows:
 ...... (1)
...... (1)

 to form one mole of
to form one mole of  and one mole of
and one mole of 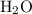 . So the number of moles of
. So the number of moles of 
 ...... (2)
...... (2)



