3. How many moles are found in 11.2 L of H2O vapor (steam) at STP?
Step by step please h...

Chemistry, 20.01.2021 09:20 Kimbeeeerly4569
3. How many moles are found in 11.2 L of H2O vapor (steam) at STP?
Step by step please help

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, monnn91351
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 17:30, kevin72937
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1


Chemistry, 23.06.2019 04:40, twinchristiansp4xhd2
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
Questions in other subjects:

Mathematics, 22.12.2019 11:31


Mathematics, 22.12.2019 11:31

Mathematics, 22.12.2019 11:31

History, 22.12.2019 11:31




Mathematics, 22.12.2019 11:31

Mathematics, 22.12.2019 11:31

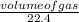
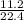 = 0.5mole
= 0.5mole 

