
Chemistry, 20.01.2021 07:30 mannablofey23
Question 1: Which of the above electron dot diagrams is WRONG and why?
A) Fluorine F because it is supposed to have 8 Valence electrons
B) Carbon C because it supposed to have 8 valence electrons
C) Nitrogen N because it is supposed to have 6 valence electrons
D) Oxygen O because it is supposed to have 6 Valence electrons
Question 2: How many valence electrons does an atom of Argon have?
A) 8
B) 7
C) 6
D) 5
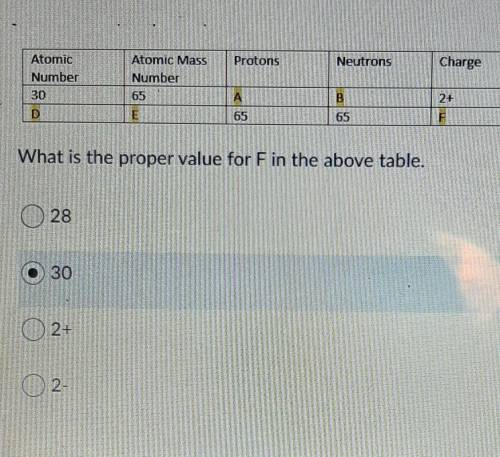
Question 3: What is the proper value for F in the above table. (Picture of table above..)
A) 28
B) 30
C) 2+
D) 2-
Question 4: What is the proper value for C in the above table. (Use the same picture as the previous question)
A) 28
B) 30
C) 32
D) 65

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, aeverettpdzrvo
The most efficient way to establish the best possible economizer position is to measure
Answers: 1

Chemistry, 21.06.2019 22:30, britotellerialuis
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

Chemistry, 22.06.2019 00:00, lilyclairehutson
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 12:30, robert7248
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2
You know the right answer?
Question 1: Which of the above electron dot diagrams is WRONG and why?
A) Fluorine F because it is...
Questions in other subjects:

Mathematics, 11.12.2020 01:00

English, 11.12.2020 01:00

Health, 11.12.2020 01:00






Mathematics, 11.12.2020 01:00




