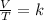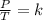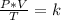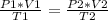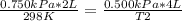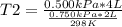Chemistry, 20.01.2021 01:50 KylaChanel4756
A sample of gas has a volume of 2.00 L and a pressure of 0.750 kPa when its
temperature is 25°C. If the volume is expanded to 4.00 L and the pressure reduced to
0.500 kPa, what must the temperature become?
379°C
397°C
379 K
397K

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, KieraKimball
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 21.06.2019 23:30, mgavyn1052
Write a paragraph that provides examples of each stage of volcanic activity, a description of the volcano, and facts about each stage.
Answers: 1

Chemistry, 22.06.2019 07:00, mayamabjishovrvq9
The variability in marine salinity between habitats does not impact the fish living there. select the best answer from the choices provided t f
Answers: 1
You know the right answer?
A sample of gas has a volume of 2.00 L and a pressure of 0.750 kPa when its
temperature is 25°C. If...
Questions in other subjects:

Mathematics, 05.04.2021 23:40




Mathematics, 05.04.2021 23:40

Mathematics, 05.04.2021 23:40