
In an experiment, potassium chlorate decomposed according to the following chemical equation.
KClO3 → KCl + O2
(Molar mass of KClO3 = 122.5 g/mol; KCl = 74.55 g/mol; O2 = 31.998 g/mol)
If the mass of potassium chlorate was 240 grams, which of the following calculations can be used to determine the mass of oxygen gas formed?
(240 × 2 × 31.998) ÷ (122.5 × 3) grams
(240 × 3 × 31.998) ÷ (122.5 × 2) grams
(240 × 2 × 122.5) ÷ (31.998 × 3) grams
(240 × 3 × 122.5) ÷ (31.998 × 2) grams

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:20, jtingley0502
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1


Chemistry, 22.06.2019 13:30, makenziehook8
Which is true of a liquid? it has a definite volume but not a definite mass. it has a definite mass but not a definite volume. it has a definite volume but not a definite shape. it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 15:00, kamkam5791
Is powdered sports drink ionic or covalent ? 10pts !
Answers: 1
You know the right answer?
In an experiment, potassium chlorate decomposed according to the following chemical equation.
KClO3...
Questions in other subjects:

Mathematics, 15.11.2020 09:40

Mathematics, 15.11.2020 09:40

Mathematics, 15.11.2020 09:40





Mathematics, 15.11.2020 09:40


Mathematics, 15.11.2020 09:50

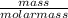
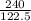
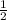 x 31.998
x 31.998


