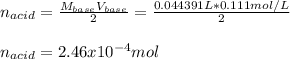Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, MickeyxX7096
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3



Chemistry, 22.06.2019 14:30, Cartucho1978
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1
You know the right answer?
An unknown diprotic acid (H2A) requires 44.391 mL of 0.111 M NaOH to completely neutralize a 0.58 g...
Questions in other subjects:

Mathematics, 14.09.2020 01:01

History, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01

Mathematics, 14.09.2020 01:01