
Chemistry, 18.01.2021 18:10 mallorytaylor279
2.In a titration, 25.00 cm3 of a solution of hydrochloric acid reacted with 18.40 cm3 of sodium hydroxide solution of concentration 0.15 mol/dm3.
The equation which represents the reaction is:
HCl + NaOH > NaCl + H2O
Calculate the concentration of hydrochloric acid in mol/dm3

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:40, petriajack8375
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3


Chemistry, 22.06.2019 04:00, eborkins
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3
You know the right answer?
2.In a titration, 25.00 cm3 of a solution of hydrochloric acid reacted with 18.40 cm3 of sodium hydr...
Questions in other subjects:

Mathematics, 22.05.2020 14:57

Mathematics, 22.05.2020 14:57

Geography, 22.05.2020 14:57


Social Studies, 22.05.2020 14:57

Mathematics, 22.05.2020 14:57


Advanced Placement (AP), 22.05.2020 14:57


English, 22.05.2020 14:57

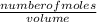 =
= 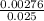 = 0.11mol/dm³
= 0.11mol/dm³

