
Chemistry, 18.01.2021 14:00 catcatscats122
Suppose your metal sample is hammered into the shape of a thin coin with a diameter of 8.62 cm. Determine the thickness (in mm) of this metal sheet. Use the measured metal mass and true denisty of your metal for this calculation.
measured metal mass: 83.956 g
true density: 8.90 g/cm3

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, lindseyklewis1p56uvi
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1

Chemistry, 22.06.2019 10:00, 2019reynolds
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1


Chemistry, 23.06.2019 15:30, Hunter1471
The gas in a sealed container has an absolute pressure of 9.25 atmospheres. if the air around the container is at standard pressure, what is the gauge pressure inside the container
Answers: 1
You know the right answer?
Suppose your metal sample is hammered into the shape of a thin coin with a diameter of 8.62 cm. Dete...
Questions in other subjects:



Mathematics, 12.07.2019 22:30

Mathematics, 12.07.2019 22:30

Mathematics, 12.07.2019 22:30

Social Studies, 12.07.2019 22:30


Mathematics, 12.07.2019 22:30


Biology, 12.07.2019 22:30

 ), measured in grams per cubic centimeter, can be represented by the following formula:
), measured in grams per cubic centimeter, can be represented by the following formula: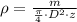 (1)
(1) - Mass, measured in grams.
- Mass, measured in grams. - Diameter, measured in centimeters.
- Diameter, measured in centimeters. - Thickness, measured in centimeters.
- Thickness, measured in centimeters.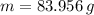 ,
,  and
and  , then the thickness of the coin is:
, then the thickness of the coin is:




