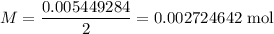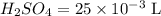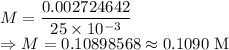Chemistry, 18.01.2021 14:00 AgarioEdit
A 25.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1322 M KOH solution. A volume of 41.22 mL of KOH is required to reach the equivalence point. What is the concentration of the unknown H2SO4 solution? Express your answer in molarity to four significant figures.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 06:50, summerjoiner
What is the volume of 3.2 moles of chlorine gas (cl2) at 295 k and 1.1 atm?
Answers: 1


Chemistry, 23.06.2019 11:30, kayabwaller4589
A) equal lines b) parallel lines c) perpendicular lines d) none of the above
Answers: 1
You know the right answer?
A 25.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1322 M KOH solu...
Questions in other subjects:

History, 20.07.2021 01:00



Mathematics, 20.07.2021 01:00

Mathematics, 20.07.2021 01:00





Mathematics, 20.07.2021 01:00

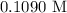
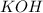 = 41.22 mL
= 41.22 mL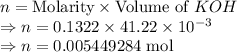
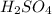
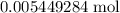 of
of