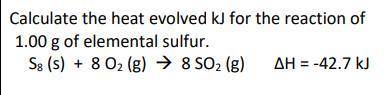Calculate the heat evolved kJ for the reaction of
1.00 g of elemental sulfur.
...


Answers: 2


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:20, emilyborland50
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d. the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 22.06.2019 10:40, rntaran2002
What is the ph of a 0.0010 m hno3? 1.0 3.0 4.0 5.0
Answers: 2

Chemistry, 22.06.2019 17:30, mwest200316
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
Questions in other subjects:


Mathematics, 26.01.2020 22:31

Mathematics, 26.01.2020 22:31



Social Studies, 26.01.2020 22:31

Mathematics, 26.01.2020 22:31