
Chemistry, 17.01.2021 18:40 sierraaasifuent
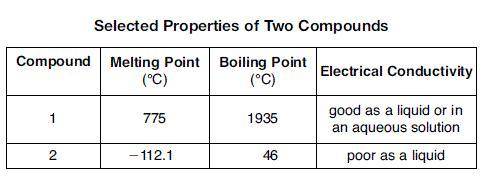
The table below shows properties of two compounds at standard pressure. Which statement classifies the two compounds? 1)Both compounds are ionic, 2)Both compounds are molecular, 3) Compound 1 is ionic, and compound 2 is molecular, 4)Compound 1 is molecular, and compound is ionic

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

You know the right answer?
The table below shows properties of two compounds at standard pressure. Which statement classifies t...
Questions in other subjects:




Mathematics, 30.07.2020 14:01



Biology, 30.07.2020 14:01



Mathematics, 30.07.2020 14:01



