
Chemistry, 16.01.2021 01:00 im1stupid1asf
This is the last question i need your help please thank you
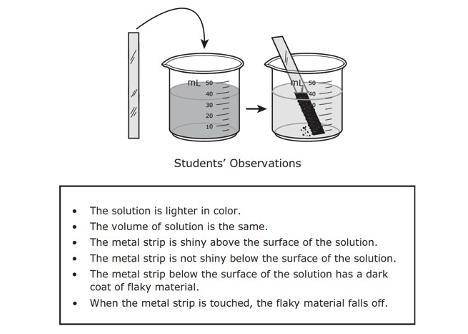
For a laboratory investigation some students put a strip of shiny metal into a beaker of blue solution and then stored the beaker on a shelf overnight. The next morning, the students recorded observations about the metal and the solution in the box below.
Based on theri observations, can the students correctly conclude that a chemical reaction occurred?
A.) No, because the material strip was still visible.
B.) Yes, because a new material of a different color formed on part of the metal strip
C.) No, because the solution stayed blue
D.) Yes, because the volume of the blue solution stayed the same

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:50, kelli151
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2


Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 06:30, jonloya264
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1
You know the right answer?
This is the last question i need your help please thank you
For a laboratory investigation some stu...
Questions in other subjects:

Mathematics, 26.09.2021 20:00

Geography, 26.09.2021 20:00

Mathematics, 26.09.2021 20:00




Mathematics, 26.09.2021 20:00

Geography, 26.09.2021 20:00


World Languages, 26.09.2021 20:00



