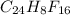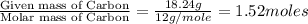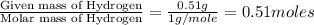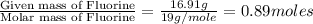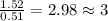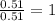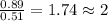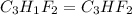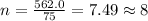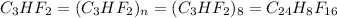Chemistry, 14.01.2021 17:00 bcarri4073
determine the empirical and molecular formula of a compound composed of 18.24 g carbon, 0.51 g hydrogen, and 16.91 g fluorine and has a molar mass 562.0 g/mol.

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
determine the empirical and molecular formula of a compound composed of 18.24 g carbon, 0.51 g hydro...
Questions in other subjects:



English, 09.01.2020 03:31


Advanced Placement (AP), 09.01.2020 03:31



Mathematics, 09.01.2020 03:31

History, 09.01.2020 03:31

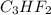 and molecular formula for the given compound is
and molecular formula for the given compound is