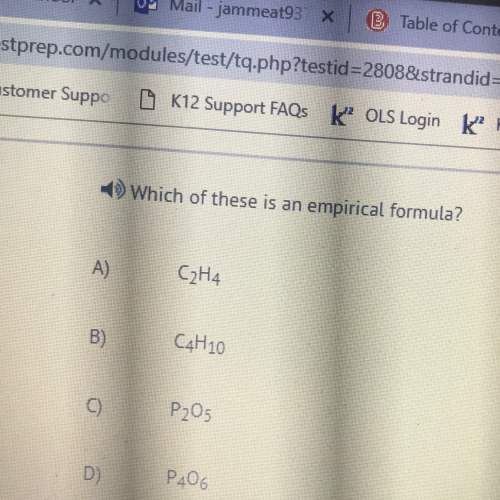
Chemistry, 13.01.2021 22:00 mikaelalcool1
Octane (C8H18) is found in gasoline. It is burned for fuel in a combustion
reaction. The unbalanced combustion reaction for octane is shown below.
C8H18 + 02 → CO2 + H2O + heat
When the reaction is balanced, how many carbon dioxide molecules are
produced for every octane molecule burned?
A. 4
o
B. 1
C. 2
O
D. 8

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 10:30, tjjjjjjjjjjjjjjjjjjj
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1
You know the right answer?
Octane (C8H18) is found in gasoline. It is burned for fuel in a combustion
reaction. The unbalanced...
Questions in other subjects:


Mathematics, 05.12.2019 09:31

Health, 05.12.2019 09:31


Mathematics, 05.12.2019 09:31

Mathematics, 05.12.2019 09:31

Chemistry, 05.12.2019 09:31



Law, 05.12.2019 09:31