
Chemistry, 13.01.2021 19:00 marygatewell385
HELP ASAP
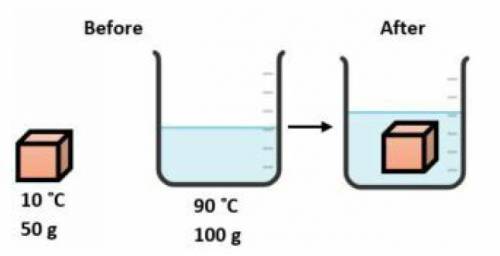
A 50.0 gram block of copper at 10.0 °C is carefully lowered into 100.0 grams of water at 90.0 °C in an insulated container as shown below. Which statement describes the transfer of heat in this system?
The water loses heat and the block gains heat until both are at the same temperature that is between 10.0°C and 90.0°C.
The water loses heat and the block gains heat until both are at the same temperature that is between 10.0°C and 90.0°C.
The water gains heat and the block loses heat until both are at the same temperature that is between 10.0°C and 90.0°C.
The water gains heat and the block loses heat until both are at the same temperature that is between 10.0°C and 90.0°C.
The water loses heat to the block until both are at 10.0°C.
The water loses heat to the block until both are at 10.0°C.
The block gains heat from the water until both are at 90.0°C.

Answers: 2


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
HELP ASAP
A 50.0 gram block of copper at 10.0 °C is carefully lowered into 100.0 grams of water at...
Questions in other subjects:



Biology, 18.12.2020 01:00



Mathematics, 18.12.2020 01:00

History, 18.12.2020 01:00


Chemistry, 18.12.2020 01:00

Mathematics, 18.12.2020 01:00



