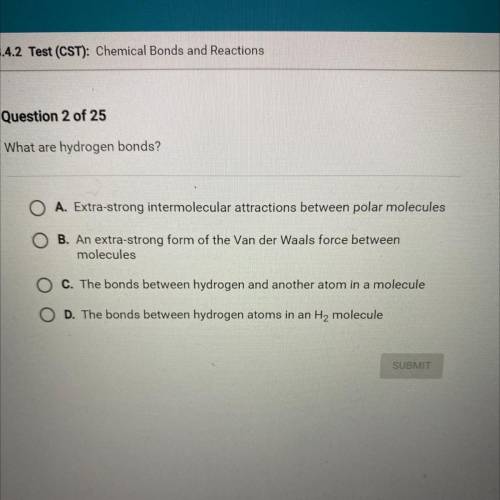
What are hydrogen bonds?
A. Extra-strong intermolecular attractions between polar molecules
B...

What are hydrogen bonds?
A. Extra-strong intermolecular attractions between polar molecules
B. An extra-strong form of the Van der Waals force between
molecules
C. The bonds between hydrogen and another atom in a molecule
D. The bonds between hydrogen atoms in an H2 molecule

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lindseysmith9522
Neils bohr believed that electrons orbited the nucleus in different energy levels, based on strong support from
Answers: 1


Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 22:30, StupidFatChipmunk
What must be in balance for temperatures to remain constant?
Answers: 1
You know the right answer?
Questions in other subjects:




English, 07.11.2020 20:20

Mathematics, 07.11.2020 20:20







