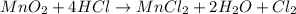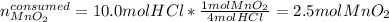Chemistry, 03.10.2019 04:40 22moneymorgan
Which is the limiting reactant when 3.00 mol of mno2 are reacted with 10.0 mol of hcl in the following equation? mno2 4hcl → mncl2 2h2o cl2

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, caeyanij
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3


Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
Which is the limiting reactant when 3.00 mol of mno2 are reacted with 10.0 mol of hcl in the followi...
Questions in other subjects:




Mathematics, 18.03.2021 22:10

Mathematics, 18.03.2021 22:10



Biology, 18.03.2021 22:10

History, 18.03.2021 22:10