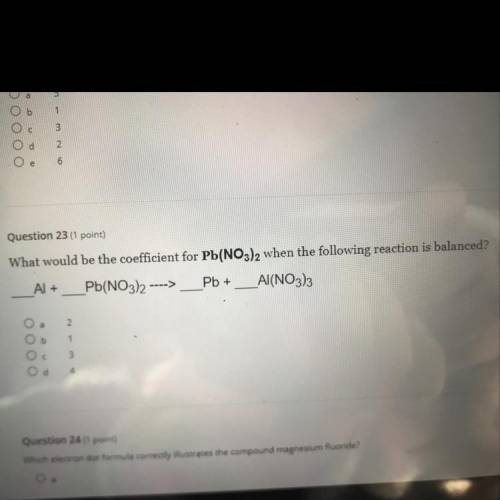What would be the coefficient for Pb(NO3)2 when the following reaction is balanced?
Al+
Pb(NO...


Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, mimithurmond03
A6.10 m nacl can be made by adding [x]g of nacl to a container and making the volume of water up to the 1.00 l line
Answers: 1

Chemistry, 21.06.2019 23:00, LarryJoeseph
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1


You know the right answer?
Questions in other subjects:

Physics, 17.04.2020 08:29

Mathematics, 17.04.2020 08:29



Mathematics, 17.04.2020 08:29


Mathematics, 17.04.2020 08:29

Physics, 17.04.2020 08:29

Mathematics, 17.04.2020 08:29

Social Studies, 17.04.2020 08:29