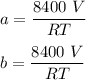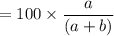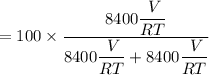Chemistry, 07.01.2021 17:30 dawsgreer4302
A mixture of ethyne gas (C2H2) and methane gas (CH4) occupied a certain volume at a total pressure of 16.8 kPa. When the sample burned, the products were CO2 gas and H2O vapor. The CO2 was collected and its pressure found to be 25.2 kPa in the same volume and at the same temperature as the original mixture. What percentage of the original mixture was methane

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, britotellerialuis
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1


Chemistry, 22.06.2019 14:30, isaiahrodriguezsm17
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1
You know the right answer?
A mixture of ethyne gas (C2H2) and methane gas (CH4) occupied a certain volume at a total pressure o...
Questions in other subjects:

Mathematics, 18.07.2019 04:30

Mathematics, 18.07.2019 04:30






Mathematics, 18.07.2019 04:30

Mathematics, 18.07.2019 04:30

Arts, 18.07.2019 04:30

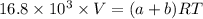
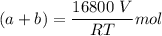 --- (1)
--- (1)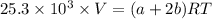
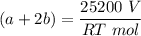 ---- (2)
---- (2)