
Chemistry, 07.01.2021 07:00 clairajogriggsk
PLEASE HELP CHEMISTRY QUESTIONS WILL GIVE BRAINLIEST
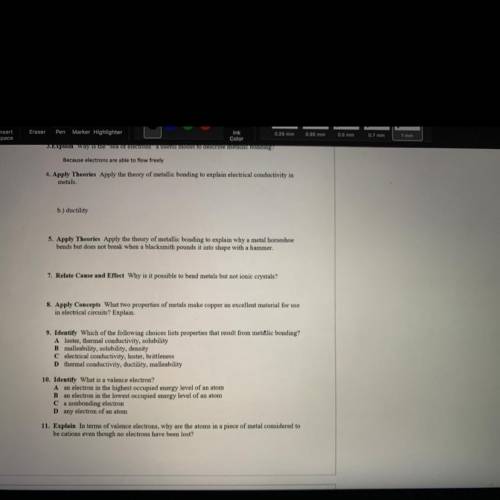
5. Apply Theories Apply the theory of metallic bonding to explain why a metal horseshoe
bends but does not break when a blacksmith pounds it into shape with a hammer.
7. Relate Cause and Effect Why is it possible to bend metals but not ionic crystals?
8. Apply Concepts What two properties of metals make copper an excellent material for use
in electrical circuits? Explain.
9. Identify Which of the following choices lists properties that result from metalic bonding?
A luster, thermal conductivity, solubility
B malleability, solubility, density
C electrical conductivity, luster, brittleness
Dthermal conductivity, ductility, malleability
10. Identify What is a valence electron?
Aan electron in the highest occupied energy level of an atom
B an electron in the lowest occupied energy level of an atom
Ca nonbonding electron
Dany electron of an atom
11. Explain In terms of valence electrons, why are the atoms in a piece of metal considered to
be cations even though no electrons have been lost?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, btcastongia
Which is a difference between molecular compounds and ionic compounds? select the correct answer below: question 5 options: molecular compounds typically form between a metal and a nonmetal, while ionic compounds typically form between nonmetals. molecular compounds result from the transfer of electrons between atoms to form ions, while ionic compounds result from the sharing of electrons between neutral atoms. molecular compounds are formed of discrete, neutral molecules, while ionic compounds are formed of large repeating arrays of opposite charges. molecular compounds have high melting points and high boiling points, while ionic
Answers: 3


Chemistry, 22.06.2019 20:00, bbyjean9974
State one important difference between a physical change and a chemical change?
Answers: 1
You know the right answer?
PLEASE HELP CHEMISTRY QUESTIONS WILL GIVE BRAINLIEST
5. Apply Theories Apply the theory of metallic...
Questions in other subjects:




English, 31.01.2022 20:30

Mathematics, 31.01.2022 20:30

Mathematics, 31.01.2022 20:30


Mathematics, 31.01.2022 20:30

Mathematics, 31.01.2022 20:30




