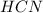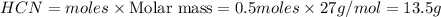Methane (CH4), ammonia (NH3), and oxygen (O2) can react to form hydrogen cyanide (HCN) and water according to this equation:
CH4+NH3+O2→HCN+H2O. A student has 8 g of methane and 10 g of ammonia in excess oxygen.
a. What is the balanced equation for this reaction?
b. Which reagent is limiting? Explain why.
c. How many grams of hydrogen cyanide will be formed?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:50, aesthetickait
How does the motion of particles in a gas change as the gas cools
Answers: 2


Chemistry, 22.06.2019 20:40, ohgeezy
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1

Chemistry, 23.06.2019 02:40, towelmearowel
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3
You know the right answer?
Methane (CH4), ammonia (NH3), and oxygen (O2) can react to form hydrogen cyanide (HCN) and water acc...
Questions in other subjects:

Mathematics, 24.11.2020 17:30


Biology, 24.11.2020 17:30

History, 24.11.2020 17:30

Social Studies, 24.11.2020 17:30


Mathematics, 24.11.2020 17:30



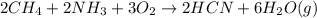
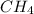 is the limiting.
is the limiting.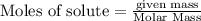
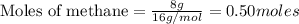
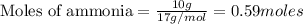
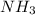
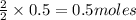 of
of