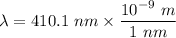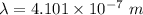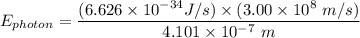The wavelength of the violet light emitted from a hydrogen atom is 410.1 nm. This light is a result of electronic transitions between the n = 5 and n = 2 energy levels. How much higher in energy is the n = 5 energy level than the n = 2 energy level?
Select one:
a. 3.000 x 108 J
b. 1.114 x 10-14 J
c. 2.436 x 10-18 J
d. 1.616 x 10-36 J
e. 4.847 x 10-19 J

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, tifftiff22
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 07:10, jasondesatnick
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

Chemistry, 22.06.2019 22:30, StupidFatChipmunk
What must be in balance for temperatures to remain constant?
Answers: 1
You know the right answer?
The wavelength of the violet light emitted from a hydrogen atom is 410.1 nm. This light is a result...
Questions in other subjects:



Mathematics, 10.02.2020 04:34

Mathematics, 10.02.2020 04:34





English, 10.02.2020 04:35

Mathematics, 10.02.2020 04:35

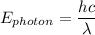
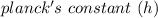 = 6.626 * 10 ^{-34} J.s
= 6.626 * 10 ^{-34} J.s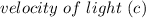 = 3.00 * 10^8 m/s
= 3.00 * 10^8 m/s