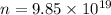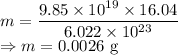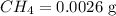Chemistry, 01.01.2021 21:30 fernandez122115
Calculate the mass in grams of each of the followinga. 5.94 x 10^20 H2O2 moleculesb. 2.8 x 10^22 SO2 moleculesc. 4.5 x 10^25 O3 moleculesd. 9.85 x 10^19 CH4 molecules

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, darkghostmist
What type of reaction fuels the processes seen here?
Answers: 2

Chemistry, 22.06.2019 19:00, nayashuntel
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1
You know the right answer?
Calculate the mass in grams of each of the followinga. 5.94 x 10^20 H2O2 moleculesb. 2.8 x 10^22 SO2...
Questions in other subjects:

Mathematics, 21.10.2020 20:01

Arts, 21.10.2020 20:01

Mathematics, 21.10.2020 20:01

Social Studies, 21.10.2020 20:01


Mathematics, 21.10.2020 20:01


History, 21.10.2020 20:01

Mathematics, 21.10.2020 20:01

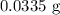
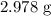
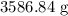
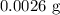
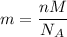
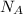 = Avogadro's number =
= Avogadro's number = 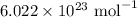
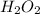 = 34.0147 g/mol
= 34.0147 g/mol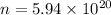
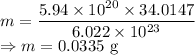
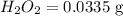
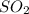 = 64.066 g/mol
= 64.066 g/mol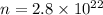
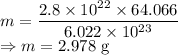
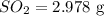
 = 48 g/mol
= 48 g/mol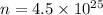
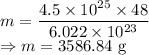
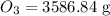
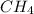 = 16.04 g/mol
= 16.04 g/mol