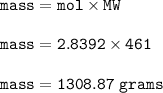Chemistry, 01.01.2021 14:00 irishvball7
18.2 mL of a 0.156 M solution of lead(II) nitrate are added to 26.2 mL of a 0.274 M solution of potassium iodide.
What is the mass of the Pbl2 precipitate formed in the reaction Pb(NO3)2 (aq) + 2 KI (aq) - Pbl2 (s) + 2 KNO3? The molar mass of Pbly is 461.0 g/mol.
Provide your answer in units of grams, with the correct number of significant digits. Enter your answer as a number only; do not include units.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, keiracoles
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1


Chemistry, 22.06.2019 20:30, demarcuswiseman
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
You know the right answer?
18.2 mL of a 0.156 M solution of lead(II) nitrate are added to 26.2 mL of a 0.274 M solution of pota...
Questions in other subjects:

English, 04.03.2020 23:46



Biology, 04.03.2020 23:46






History, 04.03.2020 23:46