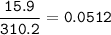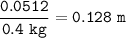Chemistry, 31.12.2020 02:30 kayleevilla
Show the calculation of the molality of a solution made by dissolving 15.9 grams of Ca3(PO4)2 in 400 grams of water.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1


Chemistry, 23.06.2019 06:40, hackman1216
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants. thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction. ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch, are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
Show the calculation of the molality of a solution made by dissolving 15.9 grams of Ca3(PO4)2 in 400...
Questions in other subjects:

Mathematics, 28.01.2020 14:04





Mathematics, 28.01.2020 14:04

Business, 28.01.2020 14:04

History, 28.01.2020 14:04

Spanish, 28.01.2020 14:04