
Step 1: (slow)
Step 2: NO3(g) + CO(g) + NO2(g) + CO2(g) (fast)
Overall: NO2(g) + CO(g) + NO(g) + CO2(g)
A two-step reaction mechanism is proposed for a gas-phase reaction, as represented above. Which of the following correctly identifies both the chemical equation for step 1 and the rate law for the overall reaction?
A) The chemical equation for step 1 is 2 NO2(g) → NO(g) + NO3(g), and the rate law is rate = (NO,].
B) The chemical equation for step 1 is NO2(g) + CO(g) + NO(g) + CO2(g), and the rate law is rate = [NO2][CO).
C) The chemical equation for step 1 is NO3(g) + 2 CO(g) + NO(g) + 2 CO2(g), and the rate law is rate = [NO2][CO).
D) The chemical equation for step 1 is NO3(g) + NO2(g) + 2 CO(g) + NO(g) + NO2(g) + 2 CO2(g), and the rate law is rate = [NO3][NO2][CO]".

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:40, loveoneonly9153
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 11:00, snowprincess99447
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 11:50, hamidaakter936848
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
You know the right answer?
Step 1: (slow)
Step 2: NO3(g) + CO(g) + NO2(g) + CO2(g) (fast)
Overall: NO2(g) + CO(g) + NO(...
Overall: NO2(g) + CO(g) + NO(...
Questions in other subjects:






Mathematics, 04.01.2020 00:31


Computers and Technology, 04.01.2020 00:31


Mathematics, 04.01.2020 00:31

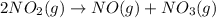 , and the rate law is
, and the rate law is ![rate = k[NO_2]^{2}](/tpl/images/1009/1195/38f22.png) .
.
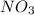 is absent in the overall reaction equation indicating that it is merely an intermediate produced in step 1 and used up in step 2.
is absent in the overall reaction equation indicating that it is merely an intermediate produced in step 1 and used up in step 2.

