
Chemistry, 26.01.2020 20:31 eldeldenali965
At room temperature, table salt is a solid and mercury is a liquid. what conclusion can you draw about the melting points of these substances

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, mayamabjishovrvq9
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2


Chemistry, 22.06.2019 20:20, Matseleng3775
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

Chemistry, 23.06.2019 00:30, Toyared
Many bird species build nests in which they raise their offspring. one of these species is known as the magnolia warbler. breeding pairs of magnolia warblers build nests out of pieces of grass and twigs. the female warbler will then lay her eggs in the nest and sit on the eggs for a few weeks until they hatch. after the offspring have hatched, the female will continue to sit on the newborn chicks to keep them warm. both the male and the female bring food for the offspring to eat until the young are mature enough to find food on their own. which of these most likely describes why birds such as the magnolia warbler build nests in which to raise their offspring? a. nest building decreases the amount of energy used by the parent birds to raise their offspring to adulthood. b. nest building increases the probability that the offspring will survive and eventually produce offspring of their own. c. nest building decreases the amount of food that the offspring require the adult birds to provide. d. nest building increases the probability of the offspring being located by airborne predators.
Answers: 1
You know the right answer?
At room temperature, table salt is a solid and mercury is a liquid. what conclusion can you draw abo...
Questions in other subjects:



Biology, 16.10.2020 20:01

Mathematics, 16.10.2020 20:01



Computers and Technology, 16.10.2020 20:01

Mathematics, 16.10.2020 20:01


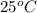 or in kelvin it is equal to 298 K.
or in kelvin it is equal to 298 K.

