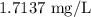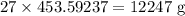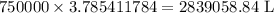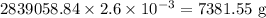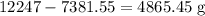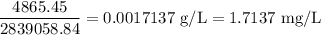Chemistry, 21.12.2020 22:20 21megoplin
Twenty-seven pounds (27 lb) of chlorine gas is used for treating 750,000 gal of water each day. The chlorine used up by the microorganisms in the water is measured to be 2.6 mg/L. What is the residual (excess) chlorine concentration in the treated water

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 22.06.2019 22:50, kanerobertrosss2213
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
Twenty-seven pounds (27 lb) of chlorine gas is used for treating 750,000 gal of water each day. The...
Questions in other subjects:


English, 26.03.2021 05:00





Mathematics, 26.03.2021 05:00

Mathematics, 26.03.2021 05:00