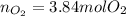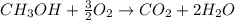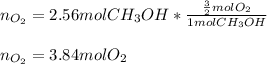Chemistry, 20.12.2020 02:40 JamierW2005
How many moles of O2 are needed to burn 2.56 moles of CH3OH?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, miamassimino
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1


Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
How many moles of O2 are needed to burn 2.56 moles of CH3OH?...
Questions in other subjects:


Mathematics, 25.06.2019 04:30

Mathematics, 25.06.2019 04:30

Mathematics, 25.06.2019 04:30




Mathematics, 25.06.2019 04:30


History, 25.06.2019 04:30