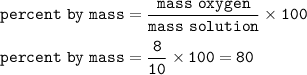Chemistry, 19.12.2020 22:20 1xXJOKERXx3
The percent by mass is the ratio of the mass of each element to the total mass of the compound expressed as a percentage. Attached is the formula. What is the percent by mass of Oxygen in 10 grams of H20 given the mass by analysis of oxygen in that sample is 8 grams. (Answer should be given with a %. i. e. if the answer were 23% you answer in the textbox would be 23%)
Percent by mass = mass of solute mass of solution x 100
help me please:((

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, ulilliareinhart2
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 06:00, giusto1894
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2


You know the right answer?
The percent by mass is the ratio of the mass of each element to the total mass of the compound expre...
Questions in other subjects:


Mathematics, 26.11.2021 21:20




History, 26.11.2021 21:20



Mathematics, 26.11.2021 21:20