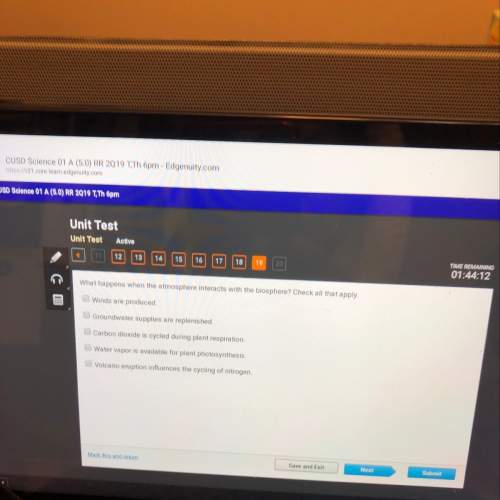
Chemistry, 17.12.2020 21:10 zaytoohypes
How does increasing the temperature affect a reaction?
OA. An increase in temperature lowers the reaction's activation energy.
OB. An increase in temperature increases the reaction rate.
OC. An increase in temperature changes equilibrium concentrations.
OD. An increase in temperature slows down the reaction.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, breannaking9734
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 14:00, emilyproce
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 22.06.2019 15:30, alaf05160
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks. energy was destroyed inside the blocks. energy was absorbed into the blocks from outside the system. energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 18:00, jeepjose58
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
How does increasing the temperature affect a reaction?
OA. An increase in temperature lowers the re...
Questions in other subjects:


English, 08.07.2020 21:01

Computers and Technology, 08.07.2020 21:01

Mathematics, 08.07.2020 21:01


Mathematics, 08.07.2020 21:01