
Chemistry, 17.12.2020 05:30 norinashanleyc
A sample of gas has a pressure of 742 torr. What is the pressure in atmospheres?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, pettygirl13
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3



Chemistry, 23.06.2019 00:00, alisonsolis155
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3
You know the right answer?
A sample of gas has a pressure of 742 torr. What is the pressure in atmospheres?...
Questions in other subjects:

English, 23.08.2021 20:50



Mathematics, 23.08.2021 20:50

Chemistry, 23.08.2021 20:50





 *
*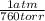 =0.976316=0.976 atm
=0.976316=0.976 atm

