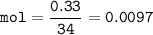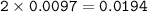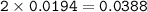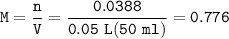Chemistry, 17.12.2020 02:30 getzperez1962
0.33 g of hydrosulfuric acid, H25, is added to 50.0 mL of water. What is the concentration (molarity) of [H301 of this solution?
H25 --> 2H +52
2H+ + 2 H20 --> 2 H30* + 2 OH
[Do not include units in your answer]

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, connienash95
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 22.06.2019 03:50, AysiaRamosLee
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 04:30, mamabates181981
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
You know the right answer?
0.33 g of hydrosulfuric acid, H25, is added to 50.0 mL of water. What is the concentration (molarity...
Questions in other subjects:

Mathematics, 28.09.2019 16:30

Mathematics, 28.09.2019 16:30

Mathematics, 28.09.2019 16:30

History, 28.09.2019 16:30



Social Studies, 28.09.2019 16:30

Biology, 28.09.2019 16:30