
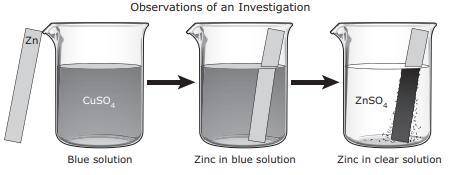
For an investigation a student poured a blue solution of CuSO4 into a beaker. The student placed a shiny, silver-colored strip of zinc metal in the solution and observed the changes. The student inferred that a chemical reaction occurred. What evidence supports this inference?
A. The CuSO4 solution turned blue when the zinc metal was added.
B. None of these
C. A dark solid formed on the zinc metal.
D. The zinc metal remained silver-colored and shiny.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, nmadrigal
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 08:40, alexisbcatlett14
Which statement can best be concluded from the ideal gas law?
Answers: 2

You know the right answer?
For an investigation a student poured a blue solution of CuSO4 into a beaker. The student placed a s...
Questions in other subjects:







Chemistry, 28.05.2021 23:20

English, 28.05.2021 23:20

Health, 28.05.2021 23:20

Mathematics, 28.05.2021 23:20



