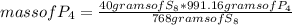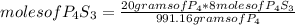The amount of P₄S₃ produced is 0.16 moles.
Explanation:
The balanced reaction is:
8 P₄ + 3 S₈ → 8 P₄S₃
By stoichiometry of the reaction (that is, the relationship between the amount of reagents and products in a chemical reaction), the following amounts of each compound participate:
P₄: 8 moles S₈: 3 moles P₄S₃: 8 moles
Being the molar mass of each compound:
P₄: 123.895 g/molS₈: 256 g/mol P₄S₃: 220.093 g/mol
then by stoichiometry of the reaction, the following amounts of reactant and product participate in the reaction:
P₄: 8 moles* 123.895 g/mol= 991.16 gS₈: 3 moles* 256 g/mol= 768 gP₄S₃: 8 moles* 220.093 g/mol= 1,760.744 g
The limiting reagent is one that is consumed first in its entirety, determining the amount of product in the reaction. When the limiting reagent is finished, the chemical reaction will stop.
In this case, you calculate the limiting reactant using the following rule of three: if by stoichiometry 768 g of S₈ react with 991.16 g of P₄, 40 g of S₈ with how much mass of P₄ will it react?
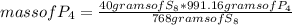
mass of P₄= 51.62 grams
But 51.62 grams of P₄ are not available, 20 grams are available. Since you have less mass than you need to react with 40 grams of S₈, P₄ will be the limiting reagent.
Then you can apply the following rule of three: if by stoichiometry 991.16 grams of P₄ form 8 moles of P₄S₃, 20 grams of P₄ will form how many moles of P₄S₃?
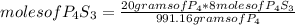
moles of P₄S₃= 0.16
The amount of P₄S₃ produced is 0.16 moles.