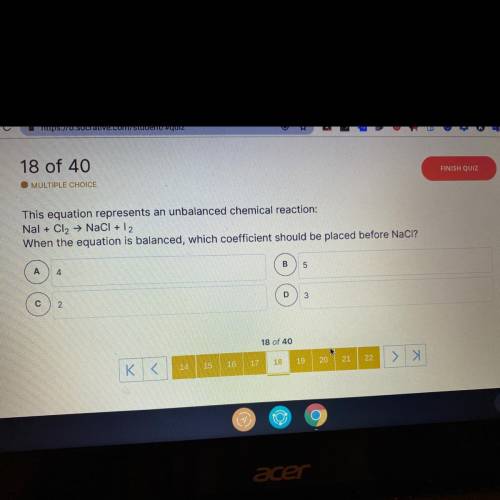
This equation represents an unbalanced chemical reaction:
Nal + Cl2 → NaCl + 12
When the equa...

Chemistry, 15.12.2020 05:40 startabull
This equation represents an unbalanced chemical reaction:
Nal + Cl2 → NaCl + 12
When the equation is balanced, which coefficient should be placed before NaCl?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, scottbrandon653
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 03:30, krharris
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 12:50, martinez6221
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 20:30, sydneip6174
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
Questions in other subjects:






Mathematics, 03.05.2021 05:50


Mathematics, 03.05.2021 05:50


Physics, 03.05.2021 05:50



