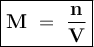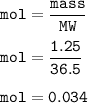Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, hdjsjfjruejchhehd
The tilt of the earth's axis of rotation is responsible for the a) ocean's tides. b) size of the moon. c) brightness of stars. d) earth’s seasons.
Answers: 1

Chemistry, 22.06.2019 23:30, adamgala3885
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1
You know the right answer?
What is the molarity of a solution prepared by dissolving 1.25g of HCl gas
into enough water to mak...
Questions in other subjects:

Social Studies, 29.01.2021 23:40


Mathematics, 29.01.2021 23:40

Mathematics, 29.01.2021 23:40

Spanish, 29.01.2021 23:40



History, 29.01.2021 23:40

Mathematics, 29.01.2021 23:40

Mathematics, 29.01.2021 23:40